By A Mystery Man Writer

The following graph denotes the variation of the compressibility factor (Z) with pressure at different temperatures for a real gas. Simply each of the curves represents an isotherm. Now, suppose w
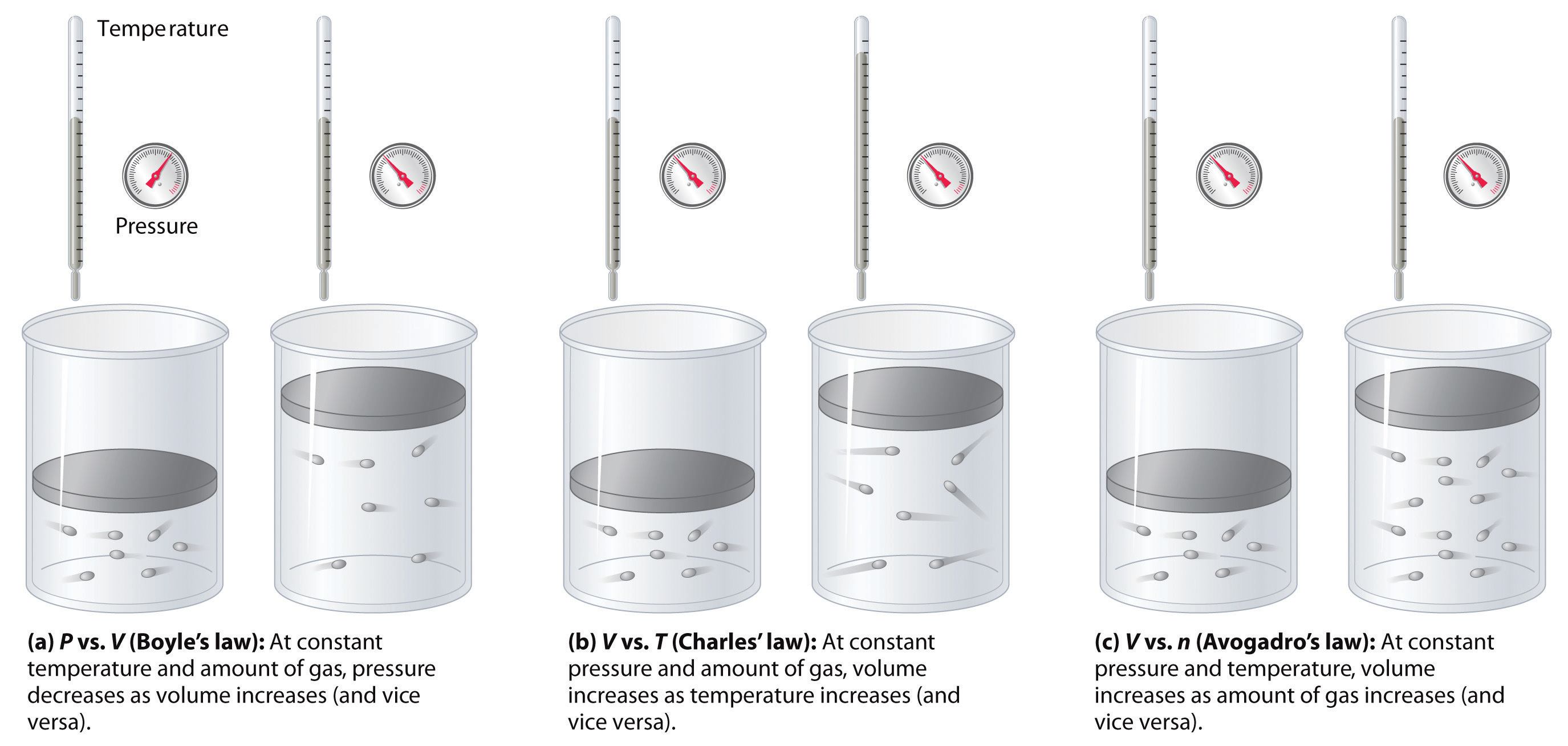
7.3: The Gas Laws - Chemistry LibreTexts

Boyle's Law Clippard Knowledgebase
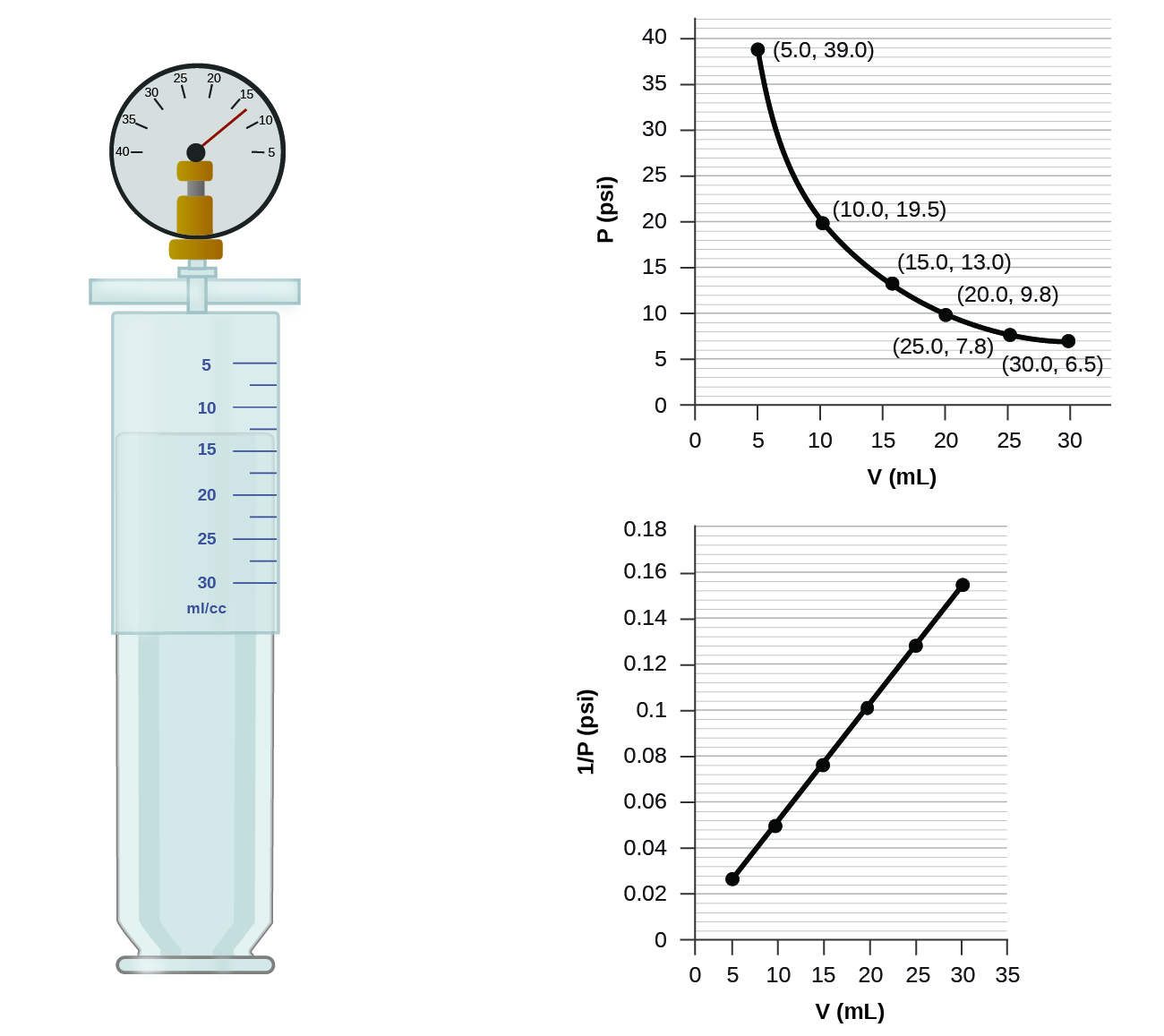
8.2 Relating Pressure, Volume, Amount, and Temperature: The Ideal Gas Law – Chemistry
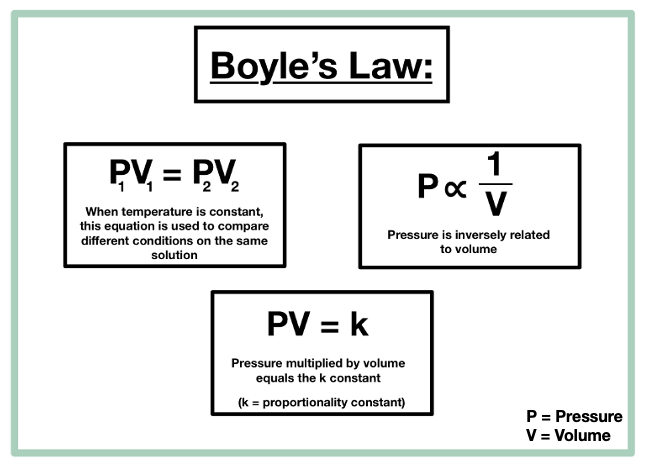
Boyle's Law — Overview & Formula - Expii

If you want to study the relationship between temperature and pressure of a gas, which factor must be held constant?

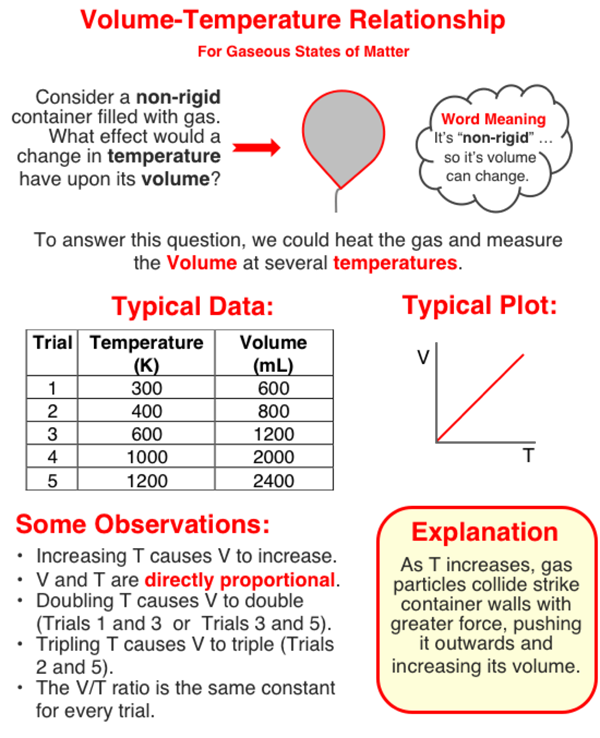
Pressure, Volume and Temperature Relationships - Chemistry Tutorial

Ideal Gas Law Formula and Examples

Gas laws Diagrams

Standard Temperature and Pressure - Ideal Gas Law
Pressure-Temperature Gas Law - Help